One aspect of research which requires substantial training, adherence, and reflection for researchers, yet gets almost no public attention, is the rules and regulations on the responsible conduct of research. In this piece I focus on the US, but many countries have ethical guidelines of their own. This piece is meant as a reflection of how far science and society have come, and while ethics in science are only as good as the scientist, I wanted to share the stringent approval and review processes that modern research must go through prior to completing any work, to ensure safety and respect for all persons and animals. Thus, please don’t read the first section and run off thinking that researchers are monsters – it is there to give you an understanding of where we ae now.
Bias and the misuse of research

If you’ve ever read “The immortal life of Henrietta Lacks”, by Rebecca Skloot, you are familiar with a high -profile case which took decades to uncover. The book examines the case of a woman, Henrietta Lacks, with cervical cancer whose cells have revolutionized medicine and research. However, doctors didn’t ask for her permission to use those cancer cells, she didn’t know they were being taken, her family didn’t know they were being used extensively in research labs around the globe, and even decades later they have not benefited from the billions of dollars of research and development profits that came about because of her cells.
History is littered with examples of cruel or nonconsensual research going back hundreds of years. Most of those involve intensive research on humans or animals without their knowledge or their consent, other examples include disregard for safety or privacy. Even decisions which appear benign but are still unethical prevent people from benefitting from their own contributions to research.
However, nearly all of these historical examples involving human subjects research are rooted in racism, sexism, and/or anti-religious or religion/ethnic cleansing. Historically and today, science has often been intentionally misconstrued to perpetuate social constructs of superiority/inferiority. Science is only a tool, and while these examples can be blamed on individuals choosing science to be the tool of their ill-intent, the historical lack of ethical guidelines, constraints, or consequences belies the failure of society to ensure equality and respect to all persons. There are numerous resources (for example, here and here) which examine these past offenses in detail, and reflect on how they led to the ethical guidelines we have in place today.
In addition to the obvious harm it could cause, not incorporating ethics into research contributes to Institutional Betrayal. This concept was coined by psychologist Dr. Jennifer Freyd, and describes the harm caused or allowed to happen by an institution, which causes psychological damage because you expect the institution to protect you. Collectively, unethical research leads to a distrust of science, researchers, and medical professionals, and can lead to science denialism.
One of the challenges to understanding ethics in scientific research is that our ethics reflect our values as a society and those values and laws change over time. You only have to read the news to see that we all have different ideas about what is acceptable to do to someone else. We should not think of everything that is permissible as also acceptable: just because you can do something, doesn’t mean you should do it. And, you can still take advantage of someone even if you are not physically harming them. Thus, ethical standards inform what we are able to research, but also why we are doing it, and how, as there may be less invasive methods available.
Importantly, ethical standards takes power away from the researcher and puts it into more objective hands. If a researcher wants to do a project, and millions of dollars of research funding, 20 research staff, and their careers are all based on the research succeeding, that puts a lot of bias in all of their thoughts and actions. Ethical review helps ensure that researchers are making good decisions before, during, and after the research.
Perception of authority and power dynamics
Americans have historically distrusted science, and this has always been encouraged by various social and political entities. This external influence on our perception of science has intensified in recent years, which has become dramatically clear in the way that people have responded to the pandemic. However, generally when people look at photos of researchers, scientists, or doctors, and they respond that they have some level of trust in them. This trust, of course, varies by gender and race and is rooted in how certain demographics have been taken advantage of previously.
There is a term called perception of authority, which can be used to describe how people ascribe authority to researchers, scientists, and doctors simply based on visual cues (like wearing a white coat). However, this perception can be incorrectly attributed to people who appear to be in that same category but are in fact not trustworthy or knowledgeable about the topics they claim to be experts in, for example, some TV personalities.
Ethical standards and review prevent researchers from intentionally or unintentionally taking advantage of participants’ willingness to say “yes” based on their perception of you being an authority, whether or not you really are. That is just one example of a power dynamic.

Research sets up a power dynamic, which is a relationship wherein one person has more power, authority, or control over another person. In research, there are a lot of ways in which that can be set up.
In addition to perception of authority, there is a perception of luck, in which participants assume they will be in the placebo group (the control group which receives no treatment to make sure the effects you see are because of the treatment and not just from the excitement of being in a research study) and dismiss concerns about potential risks. Financial incentive for participating may recruit people that really need that money and feel pressured to be int he study regardless of the risks. There is also the hope of a cure. For medical research involving obscure or rare diseases, studies may use developmental treatments and by necessity must recruit participants who suffer from this particular problem. You might be more like to participate if you assume that the treatment will cure you, or if you don’t understand that it is equally likely that you could be in a placebo group as in the treatment group. There are also people that feel pressured to consent because they have less social impact and power and feel that they can’t say ‘no’ to participation, by refusing to enroll or by withdrawing from the study at any time. Ethical regulations specifically include prisoners, children, pregnant people, and anyone without the ability to make an informed decision under special protections against power dynamics, but ethical review boards will help you identify other situations or demographics and how to lessen those power differentials.
In addition, having a study approval that rests with a committee who are in no way involved with the research can help reduce bias or harm. These standards may require researchers to be more creative in order to do less harm and find a better way to conduct that research, either by using alternative methods or fewer participants. Ethical review boards also ensure that researchers get the most out of the study, such that if some harm, even just some inconvenience, is being done to someone (human or animal), the benefits from the study are worth the cost and that judgment call is made by someone with no stake in the research. Review also ensures that the study is designed to collect as much info as possible so that it does not have be repeated.
Ethical standards also require researchers to obtain informed consent from your human participants. This includes what will be done to them during the study, what information or samples will be collected, what information (including methods) will be obtained from these samples, and what will be done with their samples or information in the future. Finally, ethical standards creates accountability for researchers’ actions by creating a paper trail, setting up oversight on the project, and creating consequences for failure to comply with regulations.
The logistics of ethics
How do we add ethical principles to our research? To summarize, you want to minimize harm to participants, be transparent about your activities and keep human participants well informed, keep excellent records and document all communications and information you share with human participants, always get Institutional approval before conducting research or collecting samples or information, and try to reduce the power dynamic by making yourself accountable for your actions. There are many guiding principles available, including some listed here provided by the NIH:
- Social and clinical value
- Scientific validity
- Fair subject selection
- Favorable risk-benefit ratio
- Independent review
- Informed consent
- Respect for potential and enrolled subjects
Institutional Animal Care and Use Committees (IACUCs)
If research involves live, vertebrate animals in some way and has a hands-on or disruptive aspect, approval from the Institutional Animal Care and Use Committee (IACUC) is required prior to starting the work. These regulations and guidelines stem from animal welfare laws and guidelines.
You should always consult with your IACUC board about your project before you have made preparations or started any work, as they should be kept apprised of research for reporting purposes and are the ones to verify if you do or do not need a formal approval. You typically don’t need formal approval if you are only observing animals and not interfering with them in any way or holding them captive, if you are collecting discarded animal products (like feathers), or if you are collecting tissue from carcasses. Keep in mind, you will need institutional biosafety approval to conduct this research if there is a specific infectious disease concern, and you need approval from your state fish and wildlife department if you are collecting samples from wild animals (even more so if the animal has a protected status). If you will be transporting biological material across state or national borders, there is another layer of training and approval before you can begin.
Each institution which performs animal research in the US is required to form at least a 5-member committee, which has to include the attending veterinarian at the institution with experience and training in the care and use of laboratory (and livestock) animals, one member from the local community, a practicing scientist experienced in research involving animals, a non-scientist, and at least one more member of any kind (usually another practicing scientist at the university).
In addition to applying to IACUC for approval for your study, you’ll need to document that everyone on the project has completed relevant training on responsible research conduct, animal handling, and the procedures you will be using. Some of that training is administered by your institution, but much of it will be performed through the Collaborative Institutional Training Initiative (CITI), which provides standardized information and training.
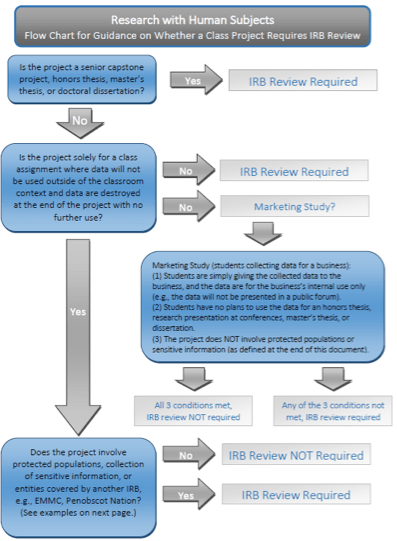
Institutional Review Boards (IRBs)
If research involves humans in some way, including surveys, approval is required from the Institutional Review Board (IRB) prior to starting the work. Even if your project ultimately does not require approval, you should always contact your IRB first, to let them know what you intend, and get their informal approval that you don’t need formal approval from the committee to do your work.
The members of the IRB committee may not have a personal, professional, or financial conflict of interest, and the federal code of regulations stipulates many guidelines about membership:
“Each IRB shall have at least five members, with varying backgrounds to promote complete and adequate review of research activities commonly conducted by the institution. The IRB shall be sufficiently qualified through the experience and expertise of its members, and the diversity of the members, including consideration of race, gender, cultural backgrounds, and sensitivity to such issues as community attitudes, to promote respect for its advice and counsel in safeguarding the rights and welfare of human subjects. In addition to possessing the professional competence necessary to review the specific research activities, the IRB shall be able to ascertain the acceptability of proposed research in terms of institutional commitments and regulations, applicable law, and standards of professional conduct and practice. * * * The IRB shall therefore include persons knowledgeable in these areas. If an IRB regularly reviews research that involves a vulnerable category of subjects, such as children, prisoners, pregnant women, or handicapped or mentally disabled persons, consideration shall be given to the inclusion of one or more individuals who are knowledgeable about and experienced in working with those subjects.”
Code of Federal Regulations, Title 21, Volume 1, Revised as of April 1, 2020, CITE: 21CFR56.107
Generally speaking, you’ll need IRB approval (and training) if you intend to publish this work or share this information widely, if you are collecting sensitive information (such as health, finances, or anything which would put the safety and wellbeing of that person at risk if it were revealed), if you are collecting (biological) samples, if you are doing anything physically or psychologically invasive, or if you are working with vulnerable populations. If you will be transporting biological material across state or national borders, there is another layer of training and approval before you can begin.
There are a number of information-gathering activities that don’t require review and approval, most of which are student projects that are part of coursework. These include interviewing one person for a biography on non-sensitive information, or interviewing multiple people on non-sensitive topics (such as asking about their favorite animal), performing a literature review or information search on non-sensitive or de-identified information, or creating science curricula.
Biosafety and chemical regulations
In addition to regulations on working with biological study subjects, there are additional health and safety regulations if your work involves anything infectious or dangerous. Institutional biosafety and chemical safety review requires researchers to describe all protocols, sample types and relative risks, and all safety and containment procedures – from the protective gear you will wear, to your sterilization or detoxification procedures, to the equipment you are using that could cause aerosolization and spread. There are yearly chemical and biosafety inventory reviews, laboratory walk-through audits, training, reporting, and equipment maintenance records that all go along with this.
Consequences
Like any good policy, responsible research is best accomplished when there are consequences and an institutional dedication to enforcement. Not only are applications and training required prior to performing the research, but there are facilities audits, reporting, and other regular check ins. Because there is no much to keep track of, review boards and enforcement are there to help researchers set up good practices and protocols ahead of time, help you stay in compliance, and correct problems before they exacerbate. Researchers who refuse to obtain permission prior to sample collection, who change their protocols without notifying review boards, who flaunt regulations, or who commit ethics violations will risk losing their funding, their job, and in severe cases, could face criminal investigations.
